
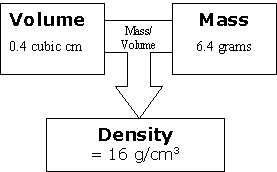
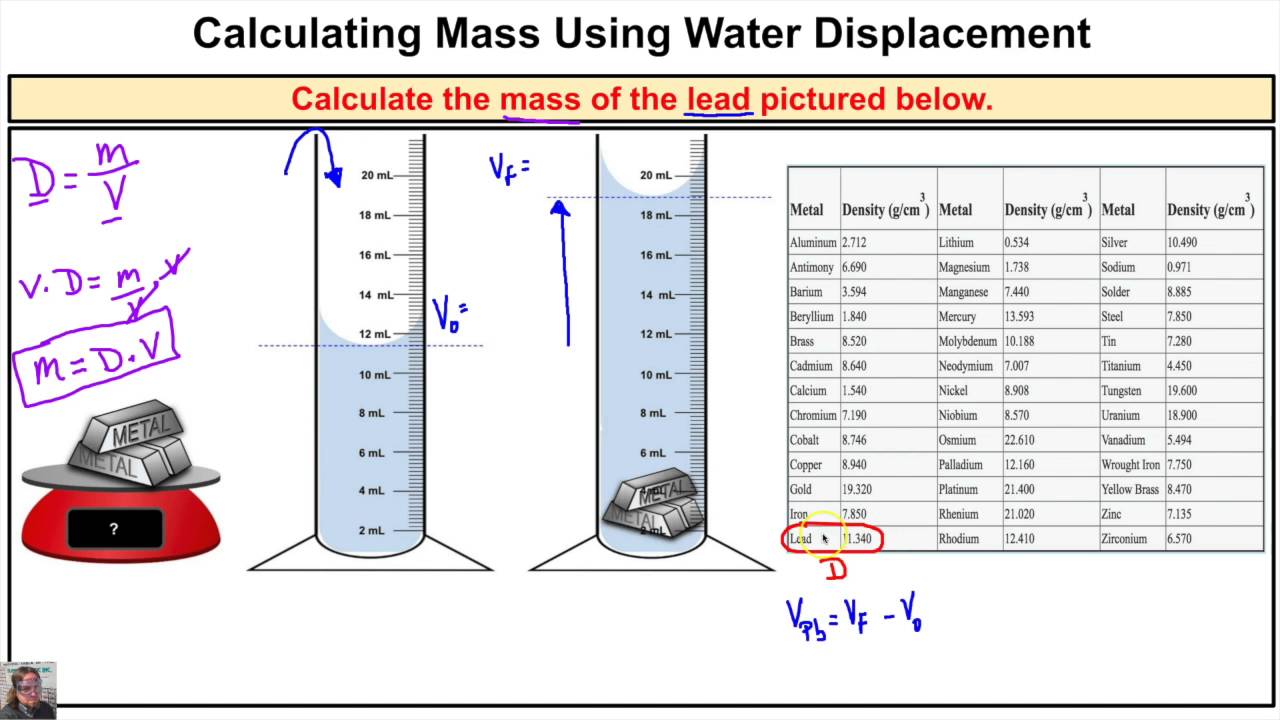
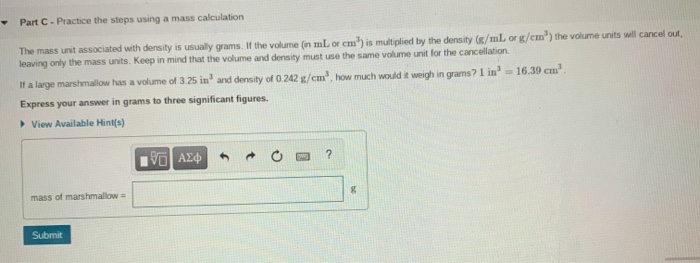
Try mixing oil and water and see how they separate into layers, with the less dense oil on top. When you mix two or more substances, the most dense substance sinks to the bottom, whilst the least dense substance is more buoyant and floats to the top. This is why you should never comment on your Auntie Mabel's sponge cake by describing it as dense: she was probably going for something a little more airy.ĭensity is also what makes things float or sink. A pound of feathers is filled with airy space, so it's not very dense at all, while a pound of lead feels much more solid, so it's more dense. For an equal amount of space it takes up (volume), lead weighs much more than the feathers (mass).ĭensity can also be thought of as how compacted or compressed a substance is. Volume-to-Weight Conversion Factors for Solid Waste This document from April 2016 provides updates to the volume-to-weight conversion factors found in the 1997 report, 'Measuring Recycling: A Guide for State and Local Governments.' Volume-to-Weight Conversion Factors for Solid Waste (pdf) (317. So if the question was, 'Which weighs more: a 500ml jug full of feathers or a 500ml jug full of lead?' then the answer would be the lead. What is density?ĭensity is the mass per volume - not just the straight-up mass. Read on to find out exactly what density is, how it works, and how to calculate density. Although the feathers and the lead both weigh exactly the same (um, a pound), their density is wildly different, and we can sometimes mentally conflate weight and density - even though they're different concepts. If you've ever been fooled by the old question, ' Which weighs more: a pound of feathers or a pound of lead?' then you wouldn't be alone.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
